 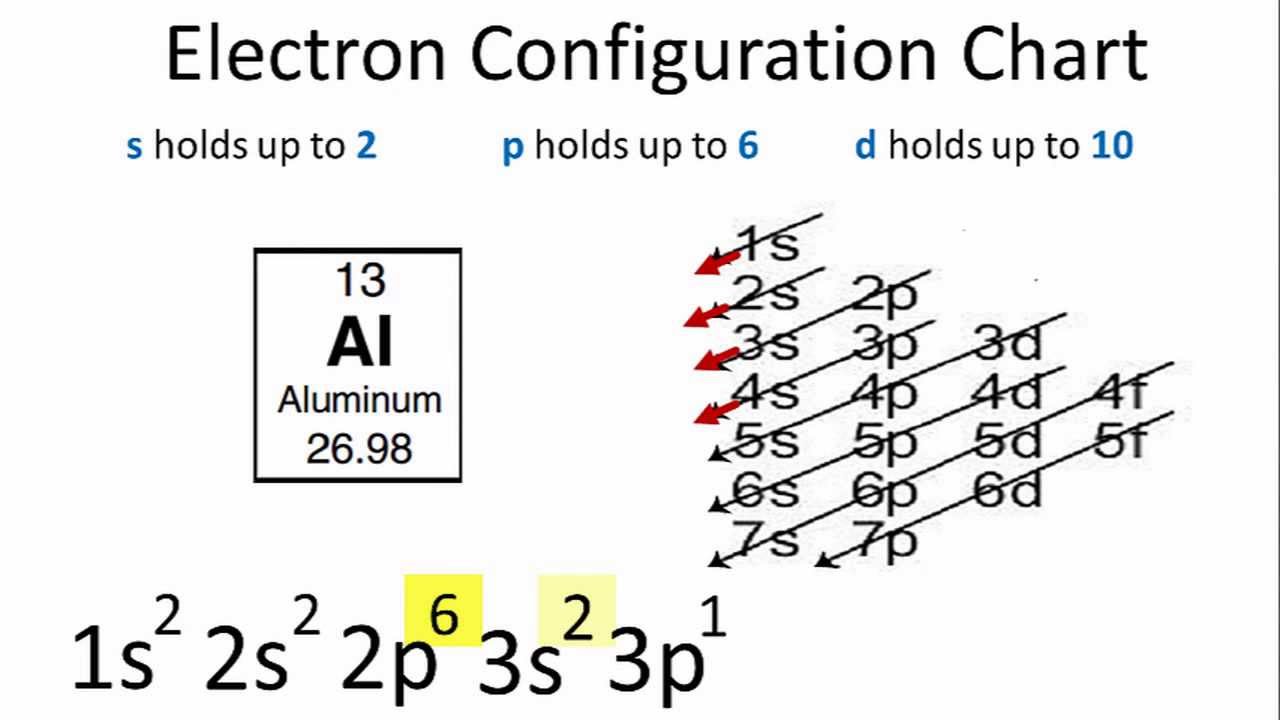
Resonance structures depict possible electronic configurations the actual configuration is a combination of the possible variations.Apply Hund's rule and justify its use to determine electron configurations for atoms in the ground state.These configurations occur in the noble gases.Electron configurations can also predict stability.The electron configuration can be written as 1s22s22p4.For example, take the electron configuration for carbon: 2 electrons will pair up in the 1s orbital, 2 electrons pair up in the 2s orbital, and the remaining 2 electrons will be placed into the 2p orbitals.Determine the electron configuration for elements and ions, identifying the relation between electron shells and subshells.Electron configurations can be used to rationalize chemical properties in both inorganic and organic chemistry.An element's electron configuration is the arrangement of the electrons in the shells.An orbital diagram helps to determine the electron configuration of an element.An atom's electrons exist in discrete atomic orbitals, and the atom's electron configuration can be determined using a set of guidelines.General Rules for Assigning Electrons to Atomic Orbitals.The electron configuration is the same as for neon and the number of nonvalence electrons is 2.The electron configuration is 1s22s2 2p6.Start by figuring out the number of nonvalence electrons, which can be determined from the electron configuration.The element sodium has the electron configuration 1s22s22p63s1.The Shielding Effect and Effective Nuclear Charge. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |


 RSS Feed
RSS Feed
